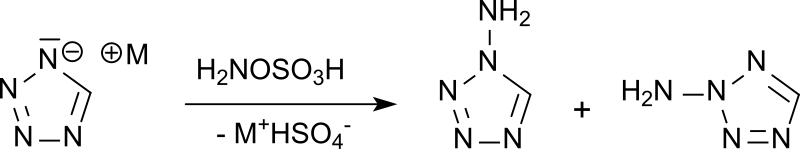
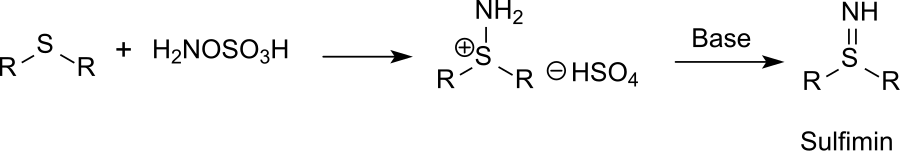
Synthesis and reactivity of heterocyclic hydroxylamine-O-sulfonates in: Heterocyclic Communications Volume 20 Issue 3 (2014)

Direct and Stereospecific Synthesis of N-H and N-Alkyl Aziridines from Unactivated Olefins Using Hydroxylamine-O-Sulfonic Acids. | Semantic Scholar

Figure 1 from Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). | Semantic Scholar

Table 1 from Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). | Semantic Scholar

Synthesis of a tricyclic lactam via Beckmann rearrangement and ring-rearrangement metathesis as key steps. - Abstract - Europe PMC
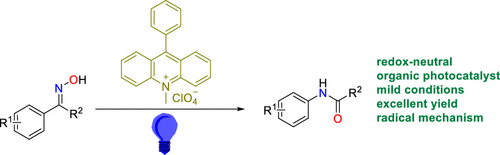
Visible-Light-Induced Beckmann Rearrangement by Organic Photoredox Catalysis.,Organic Letters - X-MOL
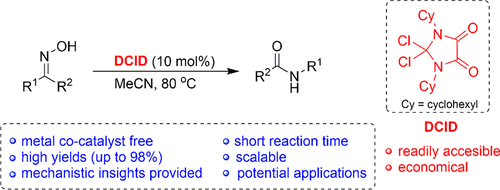
Dichloroimidazolidinedione-Activated Beckmann Rearrangement of Ketoximes for Accessing Amides and Lactams - J. Org. Chem. - X-MOL
![Synthesis of 6-alkyl analogues of the 1-azabicyclo[4.3.0]nonan-2-one system by a strategy of geminal acylation and Beckmann rearrangement - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) Synthesis of 6-alkyl analogues of the 1-azabicyclo[4.3.0]nonan-2-one system by a strategy of geminal acylation and Beckmann rearrangement - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing)](https://pubs.rsc.org/image/article/2002/p1/b108164k/b108164k-s3.gif)
Synthesis of 6-alkyl analogues of the 1-azabicyclo[4.3.0]nonan-2-one system by a strategy of geminal acylation and Beckmann rearrangement - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing)
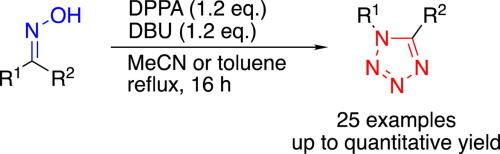
Stereospecific synthesis of 1,5-disubstituted tetrazoles from ketoximes via a Beckmann rearrangement facilitated by diphenyl phosphorazidate - Tetrahedron Lett. - X-MOL
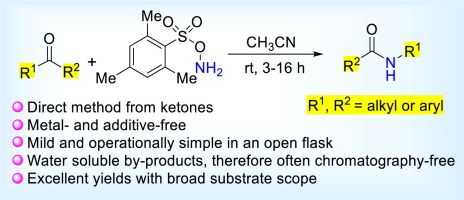
Direct Synthesis of Secondary Amides from Ketones through Beckmann Rearrangement using O-(Mesitylsulfonyl)hydroxylamine,Tetrahedron Letters - X-MOL